The Atom Challenge
The Atom Challenge
In metals, atomspack together closely, in specific arrangements, building the metal in layers. This can be seen in the diagram below which shows the atomic structure of nickel. The drawing on the left show how the atoms fit together in the individual levels, and the picture on the right shows how the layers can stack on top of each other!

When the atoms are packed as tightly as possible the layers can slide over each other - imagine each of the layers in the diagram above sliding over one another as you push or pull the structure.
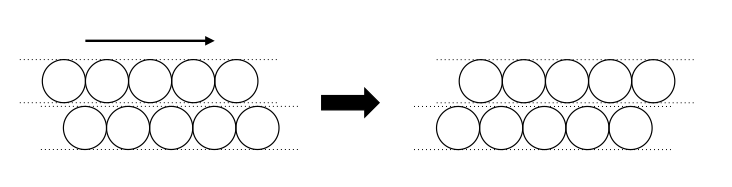
The strength and ductility (how easily it can be pulled into a wire) are due to this sliding. Whenever you deform a piece of metal, for example bending a paper clip of atoms, the planes of atoms are sliding over one another. The harder it is for the layers to slide, the stronger the metal is as it is harder to deform. So how can we make it harder for the layers to slide over one another
How can we use atoms to make them stronger?
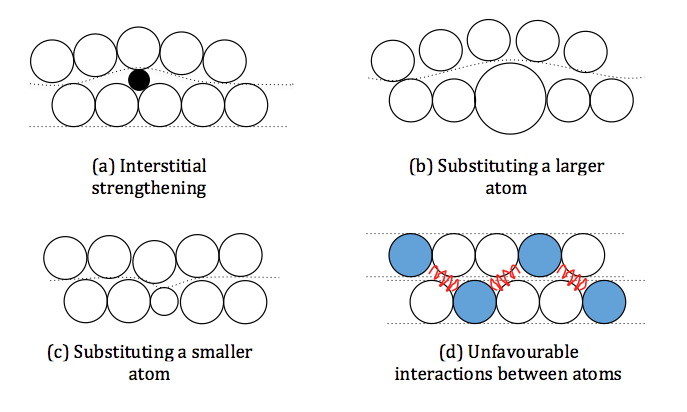
We must find a way to make it harder for the layers to slide over each other. There are four ways we can do this
a) Very small atoms, such as Carbon and Boron can fit in between the layers becoming obstacles to the sliding atoms as the layer must distort to get round them. This is known as Interstitial Strengthening.
b) Replace atoms in the layers with slightly larger ones. The atoms surrounding this larger ones must distort to accommodate it, including those in the neighbouring layers. The layers are no longer flat and so, there is no longer a smooth path for the layers to slide past.
c) Replace atoms in the layers with slightly smaller ones. The surrounding atoms move in towards the smaller atoms distorting the layers. This results in the same effect as in 2. The layers are no longer straight to slide
d) In some cases when element B prefers to be next to element A, resulting in an ordered structure where B is never next to B and A is never next to A. If the layers were to slide over each other the order would be upset and B would be next to B and A next to A. As this ruins the order it is very hard for the atoms to slide past each other
When we design the Nickel superalloy for the turbine blade we use all four to strengthen our metal. It is made up of ordered areas amongst areas where the atoms can be anywhere. Each area contains larger atoms and smaller ones to distort the layers and there are very tiny ones in between the layers.

Just doing this we have made a metal which is so strong 15 cars can hang off it without it deforming.
However, increasing the strength has downsides, we have now made it more brittle (means when it breaks it is more likely to snap - like a bar of chocolate!). There is a careful balance which means we must design a different alloy for each different requirement.
